Join us for conversations that inspire, recognize, and encourage innovation and best practices in the education profession.
Available on Apple Podcasts, Spotify, Google Podcasts, and more.

In today’s world, with populations and economies booming, the demand for energy is rising. A portfolio of different energy sources is used to meet this demand. Since there is no perfectly clean, safe, and inexpensive source of energy, the composition of this portfolio involves tradeoffs of safety, cost, and-of increasing concern-emissions of greenhouse gases such as CO2 (if you haven’t done the Carbon Cycle lab yet, we recommend you start there). In this lab, your challenge is to try to meet the world’s projected energy demand by choosing from the available energy sources while keeping atmospheric CO2under control and avoiding the particular limits and pitfalls associated with each energy source.
Unit 10: Energy Challenges // Glossary
biodiesel
A diesel-equivalent, processed fuel derived from biological sources (such as vegetable oils), that can be used in unmodified diesel-engine vehicles.
British thermal unit (Btu)
A unit of heat. One Btu is the energy required to raise one pound of water by one degree Fahrenheit at a constant pressure of one atmosphere.
clathrates
A chemical substance consisting of a lattice of one type of molecule trapping and containing a second type of molecule.
ethanol
A flammable, colorless, slightly toxic chemical compound with a distinctive perfume-like odor. Also known as ethyl alcohol, drinking alcohol, or grain alcohol, in common usage it is often referred to simply as alcohol.
fast breeder
A fast neutron reactor designed to breed fuel by producing more fissile material than it consumes.
fissile
Capable of sustaining a chain reaction of nuclear fission.
flow
The rate at which new material is added to or removed from the stock.
fuel cells
An electrochemical energy conversion device that produces electricity from external supplies of fuel (on the anode side) and oxidant (on the cathode side). Fuel cells differ from batteries in that they consume reactant, which must be replenished, while batteries store electrical energy chemically in a closed system.
gasification
A process that converts carbonaceous materials, such as coal, petroleum, petroleum coke or biomass, into carbon monoxide and hydrogen.
geothermal gradient
The rate of increase in temperature per unit depth in the Earth.
hydrates
Compounds formed by the union of water with some other substance.
hydrocarbons
Chemical compounds containing carbon and hydrogen as the principal elements. Oil is composed primarily of hydrocarbons.
inflow
General term designating the water or other fluid entering a system.
methane hydrates
Natural formations consisting of mounds of icelike material on or just below the sea floor containing large amounts of methane trapped within a lattice of icelike crystals.
non-renewable resource
A natural resource such as coal or mineral ores that is not replaceable after its removal.
oil shale
A general term applied to a fine-grained sedimentary rock containing enough organic material (called kerogen) to yield oil and combustible gas upon distillation.
ore
A mineral or an aggregate of minerals from which a valuable constituent, especially a metal, can be profitably mined or extracted.
overburden
The rock and dirt that overlie a mineral deposit and that must be removed before the mineral deposit can be extracted by surface mining.
photovoltaic
Producing an electric current as the result of light striking a metal; the direct conversion of radiant energy into electrical energy.
renewable resources
Supplies of biological organisms that can be replaced after harvesting by regrowth or reproduction of the removed species, such as seafood or timber.
source rock
A rock rich in organic matter which, if heated sufficiently, will generate oil or gas.
stock
In ecological cycles and models, the amount of a material in a certain medium or reservoir.
tar sands
Sandy deposits containing bitumen, a viscous petroleum-like material that has a high sulfur content.
thermal maturation
A process in which as rock layers press down on buried deposits, geothermal energy heats the peat and reduces its oxygen and hydrogen content, converting it to coal.
uranium enrichment
A process that results in an increase in the amount of the fissionable isotope of uranium in a given mass of uranium. Used mostly for nuclear weapons, naval propulsion, and smaller quantities for research reactors.
by Kelly Korreck

Solar power, used here on Earth, is one of the cleanest methods of energy production for use in our daily lives. However, have you ever thought about what makes the Sun so bright and powerful? The Sun is actually a very efficient nuclear reactor. Unlike the fission reactors used on Earth that split heavy atoms apart and use the leftover energy, the Sun, a fusion reactor, smashes small particles like protons together to make energy.
The Sun, like all stars, shines because of the heating of material by the nuclear fusion reactions that take place at the central core of the star. This image of the Sun, taken by TRACE (Transition Region and Coronal Explorer), shows the Sun in x-ray light.
Source: TRACE Team / NASA http://trace.lmsal.com
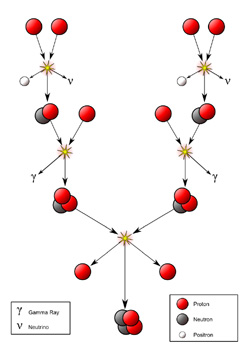 Protons and electrons make up the plasma that is in the center of our Sun. When two protons are in close proximity in this very dense area, they can combine – which gives off energy in the form of light, gamma rays, and neutrinos. The gamma rays and neutrinos heat the rest of the star as they make their way out from the center, causing the Sun to shine. The Sun has been burning hydrogen (protons) to helium (2 protons + 2 neutrons) for 5 billion years and it has enough energy to burn for another 5 billion years. When the Sun has run out of its nuclear fuel, it will eventually fissile out into a cold mass of material, but luckily not for a very long time!
Protons and electrons make up the plasma that is in the center of our Sun. When two protons are in close proximity in this very dense area, they can combine – which gives off energy in the form of light, gamma rays, and neutrinos. The gamma rays and neutrinos heat the rest of the star as they make their way out from the center, causing the Sun to shine. The Sun has been burning hydrogen (protons) to helium (2 protons + 2 neutrons) for 5 billion years and it has enough energy to burn for another 5 billion years. When the Sun has run out of its nuclear fuel, it will eventually fissile out into a cold mass of material, but luckily not for a very long time!
Fusion inside the core of the Sun starts with protons (the nucleus of the simplest form of the hydrogen atom) and progresses through several intermediate stages to end with helium. In the process, a small amount of matter is converted to energy.
Source: http://commons.wikimedia.org/wiki/
Image: FusionintheSun.png

This is the Three Mile Island Nuclear Power plant, a fission type nuclear reactor.
Source: NARA http://www.epa.gov/
The four large structures visible in the front of this nuclear power plant and along the river are the cooling towers. The nuclear reactions take place inside the reactor vessel, which holds all the fissile uranium. The uranium fuel rods are surrounded by water, which is heated by the nuclear reactions. The hot water then drives a turbine, which in turn, drives a dynamo to produce electricity. However, since there is still residual heat in the water, the water must be run through the cooling towers in order to expel more heat before being recycled into the reactor.
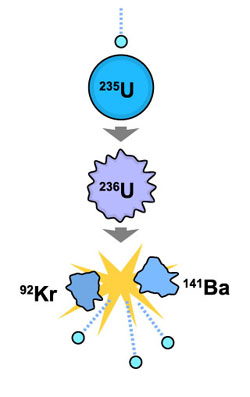
An example of a fission reaction, in which heavy uranium nuclei are split into the lighter elements, krypton and barium.
Source: http://en.wikipedia.org/wiki/Nuclear_fission
This type of fission reactor is based on the splitting of a heavy atom, such as uranium, into smaller atoms – in the process releasing the energy that was holding the atom together.
On Earth, scientists and engineers are trying to replicate the very efficient fusion processes that occur in the interior of stars, but are having great trouble overcoming engineering and plasma instabilities. Nature seems to be much more advanced!

Is an Astrophysicist in the High Energy Astrophysics Division at the Harvard-Smithsonian Center for Astrophysics. She received her Ph.D. in Space and Planetary Physics from the University of Michigan. She is part of the Hinode Solar satellite X-ray Telescope (XRT) team at the Smithsonian Astronomical Observatory. Her research interests include high energy processes in the sun and all over the universe- especially magnetic reconnection, particle heating and acceleration, and mass transfer.